Since 1833, when US army surgeon William Beaumont reported the first scientific study of a link between the gut and the brain*, science has come a long way in tracing the path of contact between the two systems.
The gut-brain axis has been known for decades now. It refers to the connections and two-way communication between the gut and the brain. This axis is crucial for normal brain and gastrointestinal (GI or gut) development and functioning.
More recently, the gut microbiota (collective term for gut microbes) has emerged as a critical regulator of the gut-brain axis. The interactions between the gut microbiota and the gut-brain axis is now referred to as the microbiota-gut-brain axis.
Health and disease implications
Studies of the microbiota-gut-brain axis have been described as a paradigm shift in neuroscience. Emerging evidence shows that gut microbiota regulates the development and many functions of the brain.
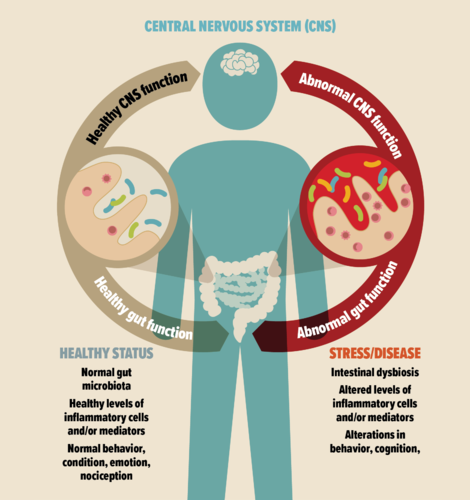
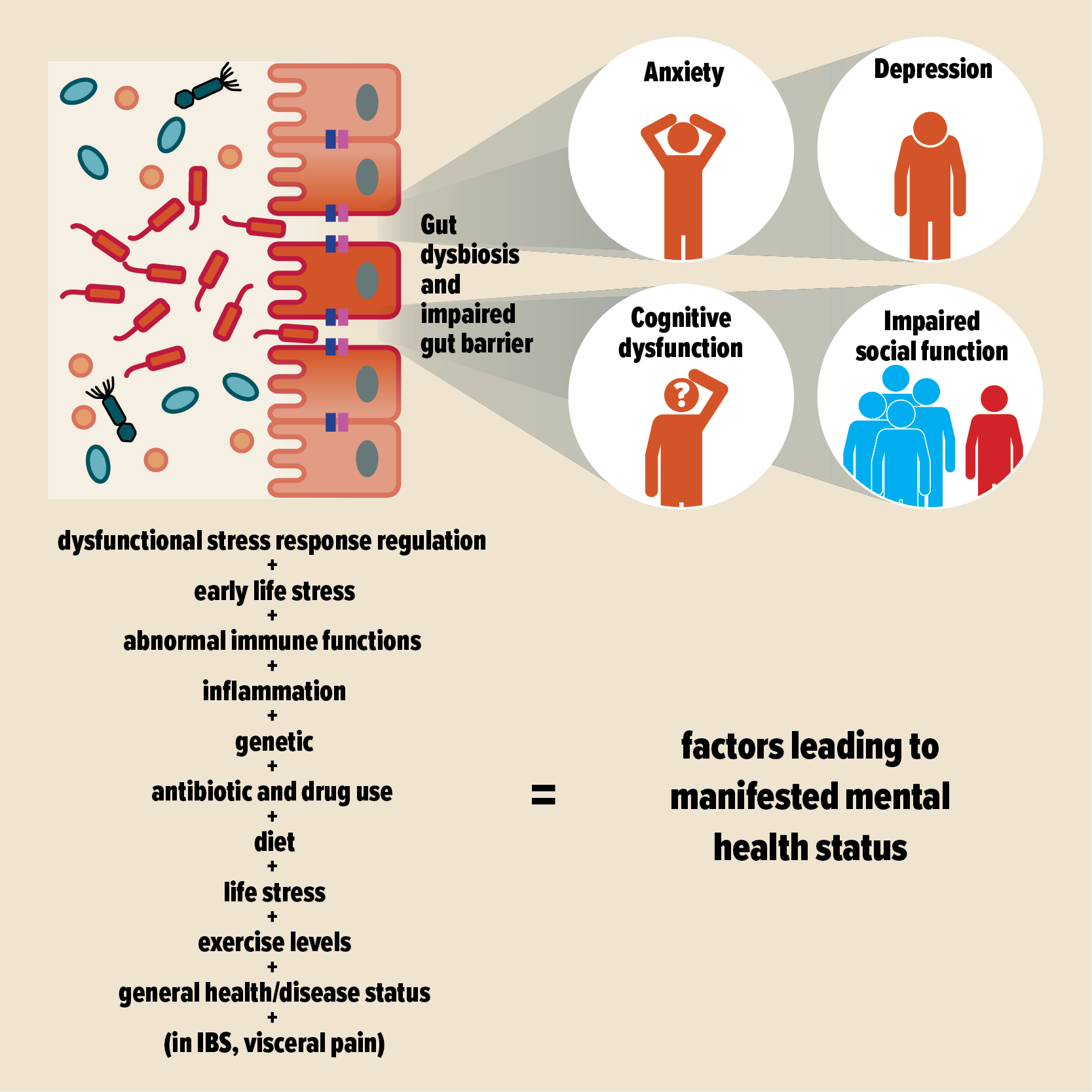
Microbiota dysbiosis (disturbance and imbalance) and leaky gut barrier, which allows entry of undigested food particles, toxic waste, and harmful bacteria into the blood circulation, affect physiological, behavioral, cognitive, and memory functions of the brain, contributing to mental health conditions and neuropsychiatric disorders.
Extensive network
The microbiota-gut-brain axis is now shown to involve multiple overlapping systems. All of which engage in constant interactions.
These systems include:
- nervous system (brain and spinal cord)
- neural system (neurons, sensors and receptors)
- neurotransmitters (serotonin and dopamine)
- endocrine system (hormones and stress response regulation)
- metabolic system (food and nutrition processing)
- immune system (immune response and gut barrier to pathogens)
Evidence from across the fields of neuroscience, gastroenterology, and microbiology support the role of gut microbes in various metabolic, gastrointestinal, neurological diseases, and their influence in brain chemistry, mental and behavioral disorders.
Communication and interactions
Communication and interactions of the microbiota-gut-brain axis follow different pathways. They include:
- direct activation of the vagus nerve
- production of alteration of neurotransmitters
- activation of immune signalling
- via microbial metabolites
- via stress-induced hormonal response
- via pro-inflammatory cells
Interactions within these systems often co-occur in the gut, which is home to approximately 500 million nerve endings and the largest concentration of immune cells in the body.
Metabolites - important players
Metabolites derived from both the digestion and microbial fermentation of dietary and nutritional components may have a significant effect on brain processes and immune responses.
For example, manipulating the composition of the gut microbiota has been shown to influence the availability and regulation of fatty acids and tryptophan. In turn, fatty acids and tryptophan can interact with the immune system, thus regulating cellular immune responses.
As an important player in the microbiota-gut-brain axis, metabolites may serve as effective targets for clinical intervention.
Figures: The orchestra plays
The illustrations are simplified view of the interplaying factors and processes. They are not attempts to explain the underlying mechanism of the processes, most of which are still unclear and being investigated.
Note:
* Source: Stress is indigestible by Stanley Coren, WellnessOptions, updated 2017, https://wellnessoptions.ca/stress-is-indigestible.html